RiskBRCA Basic
Overview
RiskBRCA Basic is our entry-level test that check for all BRCA1 and BRCA2 gene mutations related to hereditary Breast and Ovarian Cancer at the most competitive price in the market, especially designed for women at high risk, including those with a family history of Breast and/or Ovarian Cancer.

Click here to download the brochure in PDF format.
RiskBRCA Basic Basis
RiskBRCA Basic captures and direct sequences all the genomic regions for BRCA1 and BRCA2 genes, using modern Next Generation Sequencing (NGS) techniques in combination with more classical approaches of molecular genetics.
For both BRCA1 and BRCA2 genes, an average coverage of more than 300x is obtained, with which more than 99% of the regions of interest are covered above 20x. At the bioinformatic level Single Nucleotide Variant (SNVs) are detected, small indels with high sensitivity (more than 99%), as well as variants of copy number with somewhat lower sensitivity (more than 95%).
Moreover, in case of identifying variants of copy number, it is validated by Multiplex Ligation-Dependent Probe Amplification (MLPA).
Indications for use for RiskBRCA Basic
RiskBRCA Basic is indicated to detect ALL possible BRCA1 and BRCA2 mutations. RiskBRCA Basic is for high-risk women, including those with a family history of Breast and/or Ovarian Cancer.
Moreover, unlike BIOPROGNOS’ tests to help in diagnosis (such as OncoBREAST Dx, OncoLUNG Dx, OncoOVARIAN Dx, OncoPROSTATE Dx or OncoCUP Dx), RiskBRCA Basic does not need a receipt or prescription.
How to get tested
Once the purchase process is completed through this web page, you will receive a Saliva Kit at your home to take a sample of your saliva.
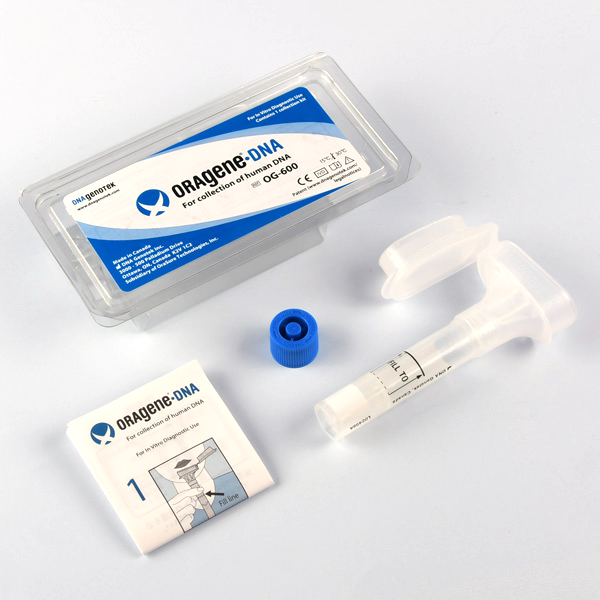
Once you receive our Saliva Kit, please just follow the instructions included and send us again with already postage paid.
After about 20 business days (depending on your location), RiskBRCA Basic results will be sent by email to the email provided during the purchase process.
References
- The BRCA1 and BRCA2 Genes and Hereditary Breast and Ovarian Cancer. CDC. Updated July 29, 2016. Accessed September 08, 2017. Available at www.cdc.gov
- Tung N, Battelli C, Allen B, et al. Frequency of mutations in individuals with breast cancer referred for BRCA1 and BRCA2 testing using next-generation sequencing with a 25-gene panel. Cancer. January 2015;121(1):25-33. Pubmed Abstract
- Pal T, Permuth-Wey J, Betts JA, et al. BRCA1 and BRCA2 mutations account for a large proportion of ovarian carcinoma cases. Cancer. December 2005;104(12):2807-16. Pubmed Abstract
- Risch HA, McLaughlin JR, Cole DE, et al. Prevalence and penetrance of germline BRCA1 and BRCA2 mutations in a population series of 649 women with ovarian cancer. Am J Hum Genet. March 2001;68(3):700-10. Pubmed Abstract
- Claus EB, Risch N, Thompson WD, et al. The calculation of breast cancer risk for women with a first degree family history of ovarian cancer. Breast Cancer Res Treat. November 1993;28(2):115-20. Pubmed Abstract
- King MC, Marks JH, Mandell JB. Breast and ovarian cancer risks due to inherited mutations in BRCA1 and BRCA2. Science. 2003;302(5645):643-6. Pubmed Abstract
- Mocci E, Milne RL, Mendez-Villamil EY, et al. Risk of pancreatic cancer in breast cancer families from the breast cancer family registry. Cancer Epidemiology Biomarkers Prev. May 2013;22(5):803-11. Pubmed Abstract
- Tai YC, Domchek S, Parmigiani G, Chen S. Breast cancer risk among male BRCA1 and BRCA2 mutation carriers. J Natl Cancer Inst. December 2007;99(23):1811-4. Pubmed Abstract
- Liede A, Karlan BY, Narod SA. Cancer risks for male carriers of germline mutations in BRCA1 or BRCA2: a review of the literature. J Clin Oncol. February 2014;22(4):735-42.
- Leongamornlert D, Mahmud N, Tymrakiewicz M, et al. Germline BRCA1 mutations increase prostate cancer risk. Br J Cancer. May 2012;106(10):1697-701.

